Direct submissions to PCI Ecology from bioRxiv.org are possible using the B2J service
Latest recommendations
| Id | Title * | Authors * | Abstract * | Picture * | Thematic fields * | Recommender | Reviewers | Submission date | |
|---|---|---|---|---|---|---|---|---|---|
24 Feb 2025

Drivers of plant-associated invertebrate community structure in West-European coastal dunesRuben Van De Walle, Maxime Dahirel, Ward Langeraert, Dries Benoit, Pieter Vantieghem, Martijn L. Vandegehuchte, François Massol and Dries Bonte https://doi.org/10.1101/2024.06.24.600350Combining Joint Species Distribution Models and multivariate techniques allows understanding biogeographical and micro-habitat community responsesRecommended by Joaquín Hortal based on reviews by Sergio Chozas, André Mira and 1 anonymous reviewer based on reviews by Sergio Chozas, André Mira and 1 anonymous reviewer
Community structure is determined by the regional species pool – which for simplicity can be assumed to be filtered through dispersal limitations, abiotic conditions, and species coexistence mechanisms (Cornell & Harrison 2014). This filtering involves macroecological constraints, such as energy and space availability, and assembly rules that determine species composition (Diamond 1975; Weiher & Keddy 1995; Guisan & Rahbek 2011; Hortal et al. 2012). But also by a series of processes that determine species distributions across scales, including biogeographical and stochastic processes (e.g., large-scale dispersal and occupancy dynamics within the landscape) and deterministic niche-based responses to abiotic and biotic conditions, which interact across scales (Soberón 2010; Hortal et al. 2010; Brousseau et al. 2018). These processes collectively determine the persistence of species assemblages within communities. It follows that, to understand the processes determining the structure of these communities it is necessary to combine methods analyse the effects of drivers acting on both species distributions and community responses. Van de Walle et al. (2025) take this integrative approach. The final revised version of their work combines multivariate techniques (in this case a RDA) and Joint SDMs to model the small-scale distribution and structure of the invertebrate communities inhabiting a series of coastal dunes in Southern England, France, Belgium and the Netherlands. The paper builds upon well-designed stratified field surveys, which allow them to identify variations at different scales, from geographical to local. These high-quality field data, together with the combination of different modelling techniques, allows them to identify both a clear biogeographical zonation in the structure of these communities, and the existence of a series of neat responses of species to the spatial structure and vigour of the tussocks created by the marram grass fixing the sand dunes. Their models also include the body size, feeding guild and phylogenetic relationships between co-occurring species, although their effects are smaller compared to those of biogeographical differences –which, arguably, are determined by differences in the species pool of each dune system, and species responses to the microhabitat conditions created by the tussocks. They can however identify a trade-off between generalist and specialist species within each community. Note that here I'm using model in the sense of tools for understanding and explaining complex ecological systems, as advocated by Levins (1966). Which is precisely what Van de Walle et al. (2025) do here. By combining techniques tailored to model species distributions and community-level responses, they (we) gain a much improved understanding of how both species pools and the spatial structure of habitats determine the composition of ecological communities. Importantly, Van de Walle et al. (2025) use this knowledge to obtain key insights about how to manage and restore these endangered habitats, thereby proving the value of this kind of integrative approaches. References Brousseau, P.-M., Gravel, D., & Handa, I. T. (2018). On the development of a predictive functional trait approach for studying terrestrial arthropods. Journal of Animal Ecology, 87(5), 1209–1220. https://doi.org/10.1111/1365-2656.12834 Cornell, H. V., & Harrison, S. P. (2014). What are species pools and when are they important? Annual Review of Ecology, Evolution, and Systematics, 45(1), 45–67. http://dx.doi.org/10.1146/annurev-ecolsys-120213-091759 Diamond, J. M. (1975). Assembly of species communities. In M. L. Cody & J. M. Diamond (Eds.), Ecology and Evolution of Communities (pp. 342–444). Harvard University Press. Guisan, A., & Rahbek, C. (2011). SESAM – a new framework integrating macroecological and species distribution models for predicting spatio-temporal patterns of species assemblages. Journal of Biogeography, 38(8), 1433–1444. https://doi.org/10.1111/j.1365-2699.2011.02550.x Hortal, J., Roura-Pascual, N., Sanders, N. J., & Rahbek, C. (2010). Understanding (insect) species distributions across spatial scales. Ecography, 33(1). https://doi.org/10.1111/j.1600-0587.2009.06428.x Hortal, J., de Marco, P., Santos, A. M. C., & Diniz-Filho, J. A. F. (2012). Integrating biogeographical processes and local community assembly. Journal of Biogeography, 39(4). https://doi.org/10.1111/j.1365-2699.2012.02684.x Levins, R. (1966). The strategy of model building in population biology. American Scientist, 54, 421–431. Soberón, J. (2010). Niche and area of distribution modeling: A population ecology perspective. Ecography, 33(1), 159–167. https://doi.org/10.1111/j.1600-0587.2009.06074.x van de Walle, R., Dahirel, M., Langeraert, W., Benoit, D., Vantieghem, P., Vandegehuchte, M. L., Massol, F., & Bonte, D. (2025). Drivers of plant-associated invertebrate community structure in West-European coastal dunes. BioRxiv, 2024.06.24.600350, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.1101/2024.06.24.600350 Weiher, E., & Keddy, P. A. (1995). Assembly rules, null models, and trait dispersion: New questions from old patterns. Oikos, 74(1), 159–164. https://doi.org/10.2307/3545686
| Drivers of plant-associated invertebrate community structure in West-European coastal dunes | Ruben Van De Walle, Maxime Dahirel, Ward Langeraert, Dries Benoit, Pieter Vantieghem, Martijn L. Vandegehuchte, François Massol and Dries Bonte | <p>The organisation of species assemblages is affected by environmental factors acting at different spatial scales. To understand the drivers behind the community structure of invertebrates associated with marram grass -the dominant dune-building ... |  | Biodiversity, Biogeography, Spatial ecology, Metacommunities & Metapopulations, Species distributions | Joaquín Hortal | 2024-06-28 10:19:36 | View | |
07 Feb 2025
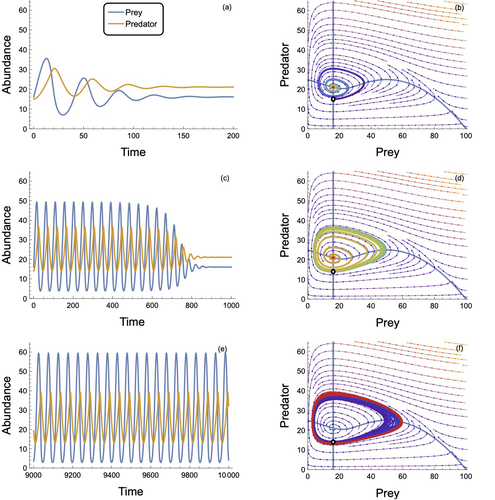
In defense of the original Type I functional response: The frequency and population-dynamic effects of feeding on multiple prey at a timeMark Novak, Kyle Edward Coblentz, John P DeLong https://doi.org/10.1101/2024.05.14.594210Revising behavioural assumptions leads to a new appreciation of an old functional response modelRecommended by Frédéric Barraquand based on reviews by Matthieu Barbier and Wojciech Uszko based on reviews by Matthieu Barbier and Wojciech Uszko
The functional response, describing the relation between predator intake rate and prey density, is a pivotal concept to understand foraging behaviour and its consequences for community dynamics. Holling (1959a) introduced three types of functional responses according to their shapes, labelled I, II and III. The type II, also known as the disc equation (Holling 1959b), has become popular among empiricists and theoreticians alike, due to its ability to describe predator intake saturation. The type III is often used to represent predator switching to other prey species when main prey density is low. Although theoretical works identify the linear functional response used in Lotka-Volterra models as a type I, Holling (1959a)’s type I model actually envisioned that at some threshold prey density, the linear increase in predator intake with prey density would give way to an upper predator intake limit, so that Holling’s type I has a rectilinear shape, with an angle joining straight lines. Ecology students can actually see this rectilinear shape reproduced in some texbooks, although not in textbook dynamical models, as they usually transition from Lotka-Volterra models to models with type II response. To many, the rectilinear shape of the original type I looks like a historical curiosity: the type II functional response accounts for intake rate saturation with a more convenient smooth function. Novak et al. (2025) turn this preconception on its head by first pedagogically showing that Holling’s original type I model can be obtained as a limit case of a variant of the celebrated type II model. The derivation follows up earlier work by Sjöberg (1980), which might be unfamiliar to readers outside aquatic ecology. The often untold assumption of the type II functional response model is that searching and handling prey are two exclusive behavioural processes, with predators that can only handle one prey item at a time. Allowing for several prey items to be handled at once while searching, until the predator reaches n prey items, the original type I functional response emerges as a limit case of the « multiprey » functional response as n goes to infinity. Interestingly, the multiprey response looks a lot like the original type I for large yet doable n. Novak et al. (2025) then proceed to look for the prevalence of such multiprey functional response shapes in a large database of functional responses (Uiterwaal et al. 2022). Combining linear type I and multiprey models (the asymptote may not always be visible), they find support for this revised type I hypothesis in about one-third of the cases. Although the type II and III models are still well supported by data, the results do suggest that linearity at low prey density may well be more frequent than one thinks. They complement this analysis by showing that larger predators relative to their prey tend to have larger n in the multiprey response. It is consistent with the hypothesis that the bigger you are relative to your prey, the more prey items you can handle at once. Finally, Novak et al. (2025) investigate the consequences of the multiprey model for community dynamics. They find overall a richer dynamical behaviour than the Lotka-Volterra type I and common parameterizations of the type II, suggesting that observed linearity in some range of prey density does not necessarily translate in simpler dynamical behaviour. Novak et al. (2025) provide here a convincing and pedagogical study showing how seemingly benign behavioural assumptions can in fact profoundly alter the perceived relevance of community dynamics models. As they conclude, their analyses have lessons for future empirical functional response work, which should not necessarily dismiss the type I model and consider perhaps variants to the classical type II and III, as well as for future theoretical analyses, which could generalize this model to multiple prey species, or relax other behavioural assumptions. References Holling, C. S. (1959a). The components of predation as revealed by a study of small-mammal predation of the European Pine Sawfly. The Canadian Entomologist, 91(5), 293-320. https://doi.org/10.4039/Ent91293-5 Holling, C. S. (1959b). Some characteristics of simple types of predation and parasitism. The Canadian Entomologist, 91(7), 385-398. https://doi.org/10.4039/Ent91385-7 Novak, M., Coblentz, K. E., & DeLong, J. P (2025). In defense of the original Type I functional response: The frequency and population-dynamic effects of feeding on multiple prey at a time. bioRxiv, ver.4 peer-reviewed and recommended by PCI Ecology https://doi.org/10.1101/2024.05.14.594210 Sjöberg, S. (1980). Zooplankton feeding and queueing theory. Ecological Modelling, 10(3-4), 215-225. https://doi.org/10.1016/0304-3800(80)90060-5 Uiterwaal, S. F., Lagerstrom, I. T., Lyon, S. R., & DeLong, J. P. (2022). FoRAGE database: A compilation of functional responses for consumers and parasitoids. Ecology, 103(7), e3706. https://doi.org/10.1002/ecy.3706 | In defense of the original Type I functional response: The frequency and population-dynamic effects of feeding on multiple prey at a time | Mark Novak, Kyle Edward Coblentz, John P DeLong | <p>Ecologists differ in the degree to which they consider the linear Type I functional response to be an unrealistic versus sufficient representation of predator feeding rates. Empiricists tend to consider it unsuitably non-mechanistic and theoret... |  | Coexistence, Community ecology, Food webs, Foraging, Population ecology, Theoretical ecology | Frédéric Barraquand | 2024-05-21 03:44:00 | View | |
24 Jan 2025
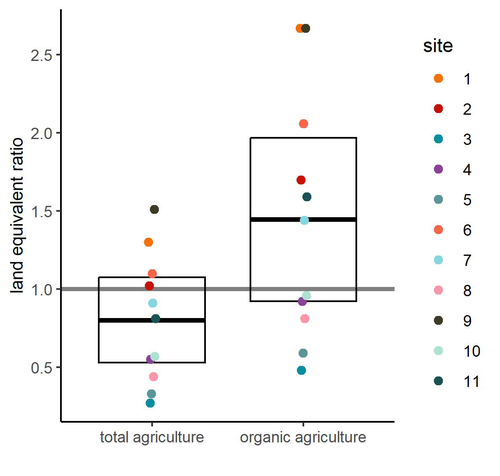
Crop productivity of Central European Permaculture is within the range of organic and conventional agriculture.Julius Reiff, Hermann F. Jungkunst, Nicole Antes, Martin H. Entling https://doi.org/10.1101/2024.09.09.611985Permaculture, a promising alternative to conventional agricultureRecommended by Aleksandra Walczyńska based on reviews by Julia Astegiano, Paulina Kramarz, Leda Lorenzo Montero and 1 anonymous reviewerAs mankind develops increasingly efficient and productive methods of agriculture and food production, we have reached a point where intensive agriculture threatens several aspects of life on Earth, negatively affecting biodiversity, carbon, nitrogen and phosphorus cycles and water reservoirs, while producing considerable amounts of greenhouse gases (Krebs and Bach, 2018). There was a need to develop farming methods that were friendly to both nature and people, producing good quality, healthy food without destroying the environment. The idea of permaculture, a concept of sustainable agriculture based on methods learned directly from nature, originated in the 1960s, invented and developed by Bruce Charles Mollison and David Holmgren (Mollison and Holmgren 1979, Mollison et al. 1991, Holmgren 2002). Although the idea of permaculture has attracted scientific interest, the representation in published studies is unbalanced in favour of positive ecological and sociological effects, with much less presence of rigorous experimental testing (Ferguson and Lovell 2014, Reiff et al. 2024a). Reiff et al. (2024b) provided the first large-scale empirical evidence of permaculture production outcomes for Central Europe. Based on results from 11 commercial permaculture sites, situated mostly in Germany but also in Switzerland and Luxembourg, the authors found that food production from permaculture sites was on average comparable to that from conventional and organic agriculture. The authors were very thorough in pointing out the issues that could potentially affect their results and which need further testing. Among these, the authors highlight the considerable variability between the 11 sites studied, which may suggest that different permacultures should differ in details according to their specificity - an interesting issue that definitely requires further study. The other factor that the authors point out that could have influenced the results and led to an underestimation of the real potential is the age of the permaculture sites. The sites from the study were relatively young, and their potential can be expected to increase with time. It is important to note that the results are mostly applicable to vegetables, as vegetable production accounted for 94% of production in the permaculture sites (followed by tree crops, 6%, and soft fruit production, 0.5%). There is therefore a need to include other types of crops produced in further studies of this type. To date, the results informing permaculture food production are urgently needed and should cover the potentially wide range of geographical regions and crops produced. The results of Reiff et al. (2025) show that rigorous testing of this issue is demanding, but the authors provide a very sound "road map" of further steps.
Literature: Ferguson R. S. and Lovell S. T. 2014. Permaculture for agroecology: design, movement, practice, and worldview. A review. Agronomy for Sustainable Development 34, 251-274. https://doi.org/10.1007/s13593-013-0181-6 Holmgren D. 2002. Permaculture: Principles & Pathways Beyond Sustainability. Holmgren Design Services, pp. 320. Krebs J. and Bach S. 2018. Permaculture – scientific evidence of principles for the agroecological design of farming systems. Sustainability 10, 3218, https://doi.org/10.3390/su10093218 Mollison B. C. and Holmgren D. 1979. Permaculture One: A Perennial Agricultural System for Human Settlements. Tagari Publications, pp. 136. Mollison B. C., Slay, R. M. and Jeeves A. 1991. Introduction to permaculture. Tagari Publications, pp. 198. Reiff J., Jungkunst H. F., Mauser K. M., Kampel S., Regending S., Rösch V., Zaller J. G. and Entling M. H. 2024a. Permaculture enhances carbon stocks, soil quality and biodiversity in Central Europe. Communications Earth & Environment 5, 305. https://doi.org/10.1038/s43247-024-01405-8 Reiff J., Jungkunst H. F., Antes N. and Entling M. H. 2024b. Crop productivity of Central European Permaculture is within the range of organic and conventional agriculture. bioRxiv, ver.2 peer-reviewed and recommended by PCI Ecology. https://doi.org/10.1101/2024.09.09.611985
| Crop productivity of Central European Permaculture is within the range of organic and conventional agriculture. | Julius Reiff, Hermann F. Jungkunst, Nicole Antes, Martin H. Entling | <p>Permaculture is a promising framework to design and manage sustainable food production systems based on mimicking ecosystems. However, there is still a lack of scientific evidence especially on the crop productivity of permaculture systems. In ... |  | Agroecology | Aleksandra Walczyńska | 2024-09-09 13:37:04 | View | |
21 Jan 2025
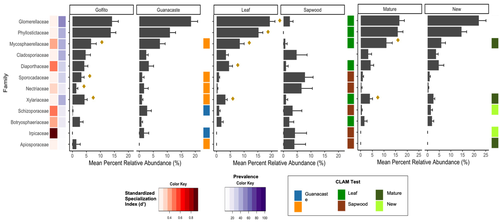
Exploring Rubiaceae fungal endophytes across contrasting tropical forests, tree tissues, and developmental stagesHumberto Castillo-González, Jason C. Slot, Stephanie Yarwood, Priscila Chaverri https://doi.org/10.1101/2024.02.13.580172The hidden diversity of fungal endophytes, associated with Rubiaceae of Costa Rican old-growth forestsRecommended by Melanie Roy based on reviews by Marion Boisseaux and 1 anonymous reviewerEndophytic fungi are expected to be hyperdiverse in tropical forests, and here is an article exploring their diversity, hidden in Rubiaceae leaves, in two old-growth forests of Costa Rica. Humberto Castillo-González et al. not only described their diversity, but also test for the impact of leaf development stage, tissue origin, and site location. They distinguish the different fungal lineages and do identify distinct indicators, showing that specialization of endophytic fungi could be related to other factors in tropical forests. This article is a great example of fungal ecology in the tropics, interacting at fine and large scale with a diversity of hosts. It also invites to discuss the high specialization observed in the tropics, and the ecology of old-growth forests in Costa Rica. References Humberto Castillo-González, Jason C. Slot, Stephanie Yarwood, Priscila Chaverri (2025) Exploring Rubiaceae fungal endophytes across contrasting tropical forests, tree tissues, and developmental stages. bioRxiv, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.1101/2024.02.13.580172 | Exploring Rubiaceae fungal endophytes across contrasting tropical forests, tree tissues, and developmental stages | Humberto Castillo-González, Jason C. Slot, Stephanie Yarwood, Priscila Chaverri | <p>Fungal endophytes play a pivotal role in tropical forest dynamics, influencing plant fitness through growth stimulation, disease suppression, stress tolerance, and nutrient mobilization. This study investigates the effects of region, leaf devel... |  | Agroecology, Biodiversity, Community ecology, Microbial ecology & microbiology, Mycology, Symbiosis | Melanie Roy | 2024-02-15 22:42:10 | View | |
20 Jan 2025
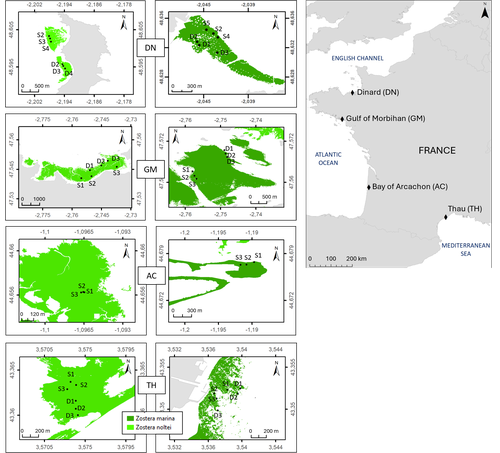
Regional and local variability in the morphometric traits of two emblematic seagrass species (Zostera marina and Zostera noltei) along the French coastÉlise Lacoste, Aurélien Boyé, Aline Blanchet-Aurigny, Nicolas Desroy, Isabelle Auby, Touria Bajjouk, Constance Bourdier, Nicolas Cimiterra, Céline Cordier, Amélia Curd, Lauriane Derrien, Élodie Foucault, Jean-Dominique Gaffet, Florian Ganthy, Loic Rigouin, Claire Rollet, Laura Soissons, Aurélien Tancray, Vincent Ouisse https://doi.org/10.5281/zenodo.10427767Importance of Scale Considerations in Understanding Seagrass DynamicsRecommended by Antoine Vernay based on reviews by Gudrun Bornette and 2 anonymous reviewers based on reviews by Gudrun Bornette and 2 anonymous reviewers
Seagrasses, particularly Zostera species, have been the subject of numerous studies due to their widespread distribution across the globe (Short et al., 2007), yet they have been in decline for several years as a result of global environmental changes (Touchette, 2007). While case studies and regional studies have been conducted, there remains a lack of information on how to scale these findings, particularly because of the heterogeneous nature of these habitats. In their study, Lacoste et al. examine the ecosystem trajectories of two Zostera species along a regional gradient spanning sites in the English Channel, the Atlantic Ocean, and the Mediterranean Sea. Their research is based on a recently published database, which offers valuable insights for comparing with other studies and serves as a resource for addressing future questions (Lacoste et al., 2024). This underscores the need for a global database to facilitate the integration of functional responses across studies, thus advancing our understanding of Zostera ecology on a larger scale. The multi-trait approach employed in their study provides a comprehensive view of population dynamics over a 1.5-year period, covering different seasons. Such studies highlight the complex responses of Zostera populations when considering environmental, seasonal, and geographical heterogeneity. Understanding these dynamics raises important questions about modeling, particularly in relation to the development of a more global database as previously mentioned. However, the review process has pointed out that the environmental data should be further refined to more rigorously support the presented results. Some statistical analyses could also benefit from improvements to ensure clearer and more explicit conclusions. These concerns are related to the challenges of sampling, the time required for such efforts, and the need to account for spatiotemporal variability. This study could serve as a foundational step for advancing our understanding of Zostera population dynamics on a global scale. In my opinion, despite the large ongoing scientific efforts, upscaling remains one of the major challenges for functional ecologists (Wood et al., 2024), particularly when plant habitats exhibit the kind of heterogeneity seen in Zostera, as demonstrated by Lacoste et al. in their work. References Élise Lacoste, Aurélien Boyé, Aline Blanchet-Aurigny, Nicolas Desroy, Isabelle Auby, Touria Bajjouk, Constance Bourdier, Nicolas Cimiterra, Céline Cordier, Amélia Curd, Lauriane Derrien, Élodie Foucault, Jean-Dominique Gaffet, Florian Ganthy, Loic Rigouin, Claire Rollet, Laura Soissons, Aurélien Tancray, Vincent Ouisse (2024) Regional and local variability in the morphometric traits of two emblematic seagrass species (Zostera marina and Zostera noltei) along the French coast. Zenodo, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.5281/zenodo.10427767 Lacoste, E., Ouisse, V., Nicolas, D., Allano, L., Auby, I., Bajjouk, T., Bourdier, C., Caisey, X., de Casamajor, M.-N., Cimiterra, N., Cordier, C., Curd, A., Derrien, L., Droual, G., Dubois, F. S., Foucault, E., Foveau, A., Gaffet, J.-D., Ganthy, F., … Blanchet-Aurigny, A. (2024). A dataset of Zostera marina and Zostera noltei structure and functioning in four sites along the French coast over a period of 18 months. https://doi.org/10.5281/zenodo.14174128 Short, F., Carruthers, T., Dennison, W., & Waycott, M. (2007). Global seagrass distribution and diversity : A bioregional model. Journal of Experimental Marine Biology and Ecology, 350(1), 3‑20. https://doi.org/10.1016/j.jembe.2007.06.012 Touchette, B. W. (2007). The biology and ecology of seagrasses. Journal of Experimental Marine Biology and Ecology, 350(1), 1‑2. https://doi.org/10.1016/j.jembe.2007.06.013 Wood, G. V., Filbee-Dexter, K., Coleman, M. A., Valckenaere, J., Aguirre, J. D., Bentley, P. M., Carnell, P., Dawkins, P. D., Dykman, L. N., Earp, H. S., Ennis, L. B., Francis, P., Franco, J. N., Hayford, H., Lamb, J. B., Ling, S. D., Layton, C., Lis, E., Masters, B., … Wernberg, T. (2024). Upscaling marine forest restoration : Challenges, solutions and recommendations from the Green Gravel Action Group. Frontiers in Marine Science, 11. https://doi.org/10.3389/fmars.2024.1364263 | Regional and local variability in the morphometric traits of two emblematic seagrass species (*Zostera marina* and *Zostera noltei*) along the French coast | Élise Lacoste, Aurélien Boyé, Aline Blanchet-Aurigny, Nicolas Desroy, Isabelle Auby, Touria Bajjouk, Constance Bourdier, Nicolas Cimiterra, Céline Cordier, Amélia Curd, Lauriane Derrien, Élodie Foucault, Jean-Dominique Gaffet, Florian Ganthy, Loic... | <p><em>Z</em>Zostera marina and Zostera noltei are two foundation species that play a crucial role in the functioning of coastal ecosystems. They occur in a wide range of environmental conditions over a large geographical area in the northern hemi... |  | Biogeography, Community ecology, Ecosystem functioning, Marine ecology, Morphometrics, Population ecology | Antoine Vernay | 2023-12-23 15:13:57 | View | |
14 Jan 2025

Delayed dichromatism in waterfowl as a convenient tool for assessing vital ratesAdrien Tableau, Iain Henderson, Sébastien Reeber, Matthieu Guillemain, Jean-François Maillard, Alain Caizergues https://doi.org/10.1101/2024.06.04.597326A cost-effective and non-invasive approach to estimating population dynamics in waterfowlRecommended by Huihuang Chen based on reviews by 2 anonymous reviewers based on reviews by 2 anonymous reviewers
This article highlights a novel non-invasive method based on the "apparent sex ratios" that exploits delayed sexual importance in waterfowl populations. Unlike traditional capture-mark-recapture (CMR) technique, which is costly, invasive, and may disturb the target species, this method infers key population dynamics, such as adult survival rate and recruitment rate, by monitoring sex ratios in counts conducted during winter. Juvenile males that resemble adult females before molting provide a unique opportunity to estimate these vital rates. This method is cost-effective, minimizes disturbance to the species, and is particularly suitable for studying protected or invasive species. References Adrien Tableau, Iain Henderson, Sébastien Reeber, Matthieu Guillemain, Jean-François Maillard, Alain Caizergues (2024) Delayed dichromatism in waterfowl as a convenient tool for assessing vital rates. bioRxiv, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.1101/2024.06.04.597326 | Delayed dichromatism in waterfowl as a convenient tool for assessing vital rates | Adrien Tableau, Iain Henderson, Sébastien Reeber, Matthieu Guillemain, Jean-François Maillard, Alain Caizergues | <p>Monitoring the number of individuals is by far the most popular strategy for studying the environmental factors that determine population dynamics and for measuring the effectiveness of management actions aimed at population recovery, control o... |  | Biological control, Conservation biology, Demography, Life history, Population ecology, Statistical ecology | Huihuang Chen | 2024-06-07 17:39:34 | View | |
14 Jan 2025

Cool topoclimates promote cold-adapted plant diversity in temperate mountain forests.Jeremy Borderieux, Emiel De Lombaerde, Karen De Pauw, Pieter Sanczuk, Pieter Vangansbeke, Thomas Vanneste, Pieter De Frenne, Jean-Claude Gégout, Josep M. Serra- Diaz https://doi.org/10.32942/X2XC8TForest microclimate in mountains and its impact on plant community: Still a question of shade, but this time it’s not coming from the canopyRecommended by Romain Bertrand based on reviews by Martin Macek and 2 anonymous reviewersRecently, microclimate has gained significant momentum [1], as evidenced by the increasing number of studies and the emergence of a dedicated scientific community coordinating research efforts [2]. Several factors underpin this trend, including advances in technology that have made microclimate monitoring [3] and ecological contextualization [4] more accessible, as well as improvements in computational methods that facilitate modeling at unprecedented scales [5]. But the growing emphasis on microclimate is primarily driven by their ecological relevance, as microclimate represent the actual climate conditions experienced by organisms [1]. This makes them more suitable than macroclimate data for understanding and predicting biodiversity responses to climate change [6]. While macroclimate data remain a common tool in ecology, they often represent generalized climatic conditions over large spatial scales. These data are typically derived from statistical models calibrated on observations collected at meteorological stations [7], which are usually located at 2 meters above the ground in open areas and at elevations compatible with human activities. Such characteristics limit the applicability of macroclimate data for understanding biodiversity responses, particularly at finer spatial scales. This is especially true in forest ecosystems, where microclimate results from the filtering of macroclimate conditions by forest habitats [8]. A simple walk in a forest during summer highlights this filtering, with the cooling effect of canopy shading and tree packing being clearly perceptible. If humans can sense these variations, they likely influence forest biodiversity. In fact, microclimates are crucial for defining the thermal niches of understory plant species [9] and understanding plant community reshuffling in response to climate warming [10]. In mountainous areas, topography adds further complexity to microclimates. The drop in temperature with elevation, known as the elevation-temperature lapse rate, is familiar, but topography also drives fine-scale variations [11]. Solar radiation hitting forest varies with aspect and hillshade, creating localized temperature differences. For example, equator-facing slopes receive more sunlight, while west-facing slopes are sunlit during the warmest part of the day. Consequently, in the northern hemisphere, southwest-facing slopes generally exhibit warmer temperatures, longer growing seasons, and shorter snow cover durations [12]. Thus, both topography and forest canopy shape the understory microclimate experienced by organisms in temperate mountainous forests. Is biodiversity more influenced by topography- or canopy-induced temperature buffering? While this question may not seem particularly interesting at first glance, understanding the underlying mechanisms of microclimate is crucial for guiding biodiversity conservation decisions in the face of climate change [13]. Poleward-facing slopes, valley bottoms, and dense canopies buffer warm episodes by creating cooler, more humid habitats that can serve as refugia for biodiversity [12]. Both buffering processes are valuable for conservation, but topography-induced buffering is generally more stable over the long term [14]. In contrast, canopy buffering is more vulnerable to human management, disturbances, and the ongoing acceleration of climate change, which is expected to drive tree mortality and lead to canopy opening [15]. Identifying the dominant buffering process in a given area is essential for mapping biodiversity refugia and fully integrating microclimate into conservation strategies. This approach can improve decision-making and actions aimed at promoting biodiversity sustainability in a warming world. The work of Borderieux and colleagues [16] offers new insights into this question through an innovative approach. They focus on temperate forests in a watershed in the Vosges Mountains, where they monitor understory temperature and inventory forest plant communities in separate samplings. Aiming to disentangle the effects of topography and forest canopy on understory temperature and its impact on plant communities, the authors deployed a network of temperature sensors using stratified sampling, balanced according to topography (elevation, aspect, and slope) and canopy cover. They then correlated mean annual temperatures (daily mean and maximum) with topographic factors and canopy cover, considering their potential interactions in a linear model. The contribution of each microclimate component was computed, and their effects on temperatures were mapped. These predictions were then confronted to floristic inventories to test whether topography- and canopy-induced temperature variations explained plant diversity and assemblages. First, the authors demonstrated that local topographic variations, which determine the amount of solar radiation reaching forests in mountainous areas, outweigh the contribution of canopy shading to understory temperatures. This result is surprising, as many previous studies have emphasized the importance of canopy buffering in shaping forest microclimate conditions [8]. However, these studies mostly focused on lowland areas or large scales, where terrain roughness has less influence. It is also unexpected because the authors observed that canopy cover varies at a smaller scale than aspect or topographic position in their study area, creating habitat heterogeneity that could reasonably drive local temperature variations. Nevertheless, the authors found that aspect, heat load, and topographic position induced more variation in microclimate than canopy filtering, significantly allowing deviations from the expected elevation-temperature lapse rate. Second, the topographic effect on understory temperature propagated to biodiversity. The authors found that topography-induced temperature offset explained plant diversity and composition, while canopy-induced temperature offset did not. Specifically, cold topoclimates harbored 30% more species than the average species richness across the inventoried plots. This increase in species richness was primarily due to an increase in cold-adapted species, highlighting the role of cold topoclimates as refugia. It is difficult to assess the extent to which these results are influenced by the specific forest context of the study area chosen by the authors, as there is no clear consensus in previous research regarding the role of topoclimate. For example, Macek et al. (2019) [17] highlighted the predominance of topography in controlling temperature and, consequently, forest community structure in the Czech Republic, while Vandewiele et al. (2023) [18] demonstrated the dominance of canopy control in the German Alps. The forest conditions investigated by Borderieux et al. (2025) were narrow, as they focused mainly on closed forests (more than 80% of the study area and sampling sites exhibiting canopy cover greater than 79%). Given that the canopy buffering effect on temperature increases with canopy cover until plateauing at around 80% [19], this may explain why the authors did not find a strong contribution from the canopy. Nevertheless, the methodology and case presented in their study are both innovative and applicable to other mountainous regions. The work of Borderieux et al. (2025) deserves attention for highlighting a frequently overlooked component of forest microclimate, as canopy filtering is typically regarded as the dominant driver. Topoclimate is a critical factor to consider when protecting cold-adapted forest species in the context of global warming, especially since topographic features are less subject to change than canopy cover. Future research should aim to test this hypothesis across a broader range of forest and topography conditions to identify general patterns, as well as assess the long-term effectiveness of these topographic refugia for biodiversity. It remains unclear whether the cooling effect provided by topoclimate will be sufficient to stabilize climate conditions despite the expected acceleration of climate warming in the coming decades, and whether it will be able to preserve cold-adapted species, which are among the most unique but threatened components of mountain biodiversity. References [1] Kemppinen, J. et al. Microclimate, an important part of ecology and biogeography. Global Ecology and Biogeography 33, e13834 (2024). https://doi.org/10.1111/geb.13834 [2] Lembrechts, J. J. et al. SoilTemp: A global database of near-surface temperature. Global Change Biology 26, 6616–6629 (2020). https://doi.org/10.1111/gcb.15123 [3] Wild, J. et al. Climate at ecologically relevant scales: A new temperature and soil moisture logger for long-term microclimate measurement. Agricultural and Forest Meteorology 268, 40–47 (2019). https://doi.org/10.1016/j.agrformet.2018.12.018 [4] Zellweger, F., Frenne, P. D., Lenoir, J., Rocchini, D. & Coomes, D. Advances in Microclimate Ecology Arising from Remote Sensing. Trends in Ecology & Evolution 34, 327–341 (2019). https://doi.org/10.1016/j.tree.2018.12.012 [5] Haesen, S. et al. ForestTemp – Sub-canopy microclimate temperatures of European forests. Global Change Biology 27, 6307–6319 (2021). https://doi.org/10.1111/gcb.15892 [6] Lembrechts, J. J. et al. Comparing temperature data sources for use in species distribution models: From in-situ logging to remote sensing. Global Ecology and Biogeography 28, 1578–1596 (2019). https://doi.org/10.1111/geb.12974 [7] Fick, S. E. & Hijmans, R. J. WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. International Journal of Climatology 37, 4302–4315 (2017). https://doi.org/10.1002/joc.5086 [8] De Frenne, P. et al. Global buffering of temperatures under forest canopies. Nat Ecol Evol 3, 744–749 (2019). https://doi.org/10.1038/s41559-019-0842-1 [9] Haesen, S. et al. Microclimate reveals the true thermal niche of forest plant species. Ecology Letters 26, 2043–2055 (2023). https://doi.org/10.1111/ele.14312 [10] Zellweger, F. et al. Forest microclimate dynamics drive plant responses to warming. Science 368, 772–775 (2020). https://doi.org/10.1126/science.aba6880 [11] Rolland, C. Spatial and Seasonal Variations of Air Temperature Lapse Rates in Alpine Regions. Journal of climate, 16(7), 1032-1046 (2003). https://doi.org/10.1175/1520-0442(2003)016%3C1032:SASVOA%3E2.0.CO;2 [12] Rita, A. et al. Topography modulates near-ground microclimate in the Mediterranean Fagus sylvatica treeline. Sci Rep 11, 1–14 (2021). https://doi.org/10.1038/s41598-021-87661-6 [13] Bertrand, R., Aubret, F., Grenouillet, G., Ribéron, A. & Blanchet, S. Comment on “Forest microclimate dynamics drive plant responses to warming”. Science 370, eabd3850 (2020). https://doi.org/10.1126/science.abd3850 [14] Hylander, K., Greiser, C., Christiansen, D. M. & Koelemeijer, I. A. Climate adaptation of biodiversity conservation in managed forest landscapes. Conservation Biology 36, e13847 (2022). https://doi.org/10.1111/cobi.13847 [15] McDowell, N. G. & Allen, C. D. Darcy’s law predicts widespread forest mortality under climate warming. Nature Clim Change 5, 669–672 (2015). https://doi.org/10.1038/nclimate2641 [16] Borderieux, J. et al. Cool topoclimates promote cold-adapted plant diversity in temperate mountain forests. Ecoevorxiv, ver. 3( 2024). Peer-reviewed and recommended by PCI Ecology https://doi.org/10.32942/X2XC8T [17] Macek, M., Kopecký, M. & Wild, J. Maximum air temperature controlled by landscape topography affects plant species composition in temperate forests. Landscape Ecol 34, 2541–2556 (2019). https://doi.org/10.1007/s10980-019-00903-x [18] Vandewiele, M. et al. Mapping spatial microclimate patterns in mountain forests from LiDAR. Agricultural and Forest Meteorology 341, 109662 (2023). https://doi.org/10.1016/j.agrformet.2023.109662 [19] Zellweger, F. et al. Seasonal drivers of understorey temperature buffering in temperate deciduous forests across Europe. Global Ecology and Biogeography 28, 1774–1786 (2019). https://doi.org/10.1111/geb.12991
| Cool topoclimates promote cold-adapted plant diversity in temperate mountain forests. | Jeremy Borderieux, Emiel De Lombaerde, Karen De Pauw, Pieter Sanczuk, Pieter Vangansbeke, Thomas Vanneste, Pieter De Frenne, Jean-Claude Gégout, Josep M. Serra- Diaz | <p>Climate strongly influences the composition and diversity of forest plant communities. Recent studies have highlighted the role of tree canopies in shaping understory thermal conditions at small spatial scales (i.e. microclimate), especially in... |  | Biodiversity, Climate change, Community ecology, Spatial ecology, Metacommunities & Metapopulations, Terrestrial ecology | Romain Bertrand | 2024-07-05 00:17:37 | View | |
06 Jan 2025

Using informative priors to account for identifiability issues in occupancy models with identification errorsCélian Monchy, Marie-Pierre Etienne, Olivier Gimenez https://doi.org/10.1101/2024.05.07.592917Accounting for false positives and negatives in monitoring data from sensor networks and eDNARecommended by Damaris Zurell based on reviews by Saoirse Kelleher, Jonathan Rose and 2 anonymous reviewersBiodiversity monitoring increasingly relies on modern technologies such as sensor networks and environmental DNA. These high-throughput methods allow biodiversity assessments with unprecedented detail and are especially useful to detect rare and secretive species that are otherwise difficult to observe with traditional survey-based methods. False negatives through imperfect detection are a typical problem in survey data and depend on intrinsic characteristics of the species, site characteristics of the survey site as well as survey characteristics (Guillera 2017). While imperfect detection might be reduced in modern sensor data and eDNA data, also these types of data are by no means error-free and may bare other challenges. In particular, the bioinformatics and image classification approaches used for species identification from these data can induce a higher rate of false positives than would be expected in expert-based survey data (Hartig et al. 2024). Occupancy models (or occupancy-detection models) have been widely used to map species distributions by fitting a hierarchical model that estimates the paramaters of both the species-environment relationship and an observation submodel. They account for false negatives by inferring detectability from the detection history of a survey location, for example from replicate visits or multiple observers (Guillera 2017). These basic occupancy-detection models assume no false positive errors in the data. Other authors have proposed extensions for false positives that typically rely on unambiguous (known truth) information for some sites or observations (Chambert et al. 2015). In their preprint, Monchy et al. (2024) propose an extension of classic occupancy models that considers a two-step observation process modelling the detection probability at occupied sites and the associated identification probability, separated into the true positive identification rate and the true negative identification rate. Using a simulation approach, the authors compare the effectiveness of a frequentist (maximum likelihood-based) and Bayesian approach for parameter estimation and identifiability, and additionally test the effectiveness of different priors (from non-informative to highly informative). Results of the maximum-likelihood approach indicated biased parameter estimates and identifiability problems. In the Bayesian approach, inclusion of prior information greatly reduces biases in parameter estimates, especially in detection and positive identification rate. Importantly, informative priors for the identification process are a by-product of the classifiers that are developed for processing the eDNA data or sensor data. For example, species identification from acoustic sensors is based on image classifiers trained on labelled bird song spectrograms (Kahl et al. 2021) and as part of the evaluation of the classifier, the true positive rate (sensitivity) is routinely being estimated and could thus be readily used in occupancy models accounting for false positives. Thus, the approach proposed by Monchy et al. (2024) is not only highly relevant for biodiversity assessments based on novel sensor and eDNA data but also provides very practical solutions that do not require additional unambiguous data but recycle data that are already available in the processing pipeline. Applying their framework to real-world data will help reducing biases in biodiversity assessments and through improved understanding of the detection process it could also help optimising the design of sensor networks. References Thierry Chambert, David A. W. Miller, James D. Nichols (2015), Modeling false positive detections in species occurrence data under different study designs. Ecology, 96: 332-339. https://doi.org/10.1890/14-1507.1 Gurutzeta Guillera-Arroita (2017) Modelling of species distributions, range dynamics and communities under imperfect detection: advances, challenges and opportunities. Ecography, 40: 281-295. https://doi.org/10.1111/ecog.02445 Florian Hartig, Nerea Abrego, Alex Bush, Jonathan M. Chase, Gurutzeta Guillera-Arroita, Mathew A. Leibold, Otso Ovaskainen, Loïc Pellissier, Maximilian Pichler, Giovanni Poggiato, Laura Pollock, Sara Si-Moussi, Wilfried Thuiller, Duarte S. Viana, David I. Warton, Damaris Zurell D, Douglas W. Yu (2024) Novel community data in ecology - properties and prospects. Trends in Ecology & Evolution, 39: 280-293. https://doi.org/10.1016/j.tree.2023.09.017 Stefan Kahl, Connor M. Wood, Maximilian Eibl, Holger Klinck (2021) BirdNET: A deep learning solution for avian diversity monitoring. Ecological Informatics, 61: 101236. https://doi.org/10.1016/j.ecoinf.2021.101236 Célian Monchy, Marie-Pierre Etienne, Olivier Gimenez (2024) Using informative priors to account for identifiability issues in occupancy models with identification errors. bioRxiv, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.1101/2024.05.07.592917 | Using informative priors to account for identifiability issues in occupancy models with identification errors | Célian Monchy, Marie-Pierre Etienne, Olivier Gimenez | <p> Non-invasive monitoring techniques like camera traps, autonomous recording units and environmental DNA are increasingly used to collect data for understanding species distribution. These methods have prompted the development of statistica... |  | Statistical ecology | Damaris Zurell | 2024-05-11 12:04:10 | View | |
17 Dec 2024
Long-term survey of intertidal rocky shore macrobenthic community metabolism and structure after primary successionAline Migné, François Bordeyne, Dominique Davoult https://hal.science/hal-0434775610 years of primary succession in intertidal communities: specific and functional changesRecommended by Gudrun Bornette based on reviews by Thomas Guillemaud and John Griffin based on reviews by Thomas Guillemaud and John Griffin
This very interesting article describes the changes taking place on artificial substrates placed in an intertidal zone. The study presents an ambitious data set and demonstrates the importance of long-term monitoring to identify community dynamics. In summary, in the short term, the authors observe a phase of complexification of the communities and a peak in productivity, but after a few years, the macro-algae disappear in favour of limpets, a situation that persists after 10 years of monitoring. Monitoring over the short term would lead to an erroneous analysis of the succession patterns and dynamics of the communities, which has important consequences in terms of the recolonisation of artificial substrates in the marine environment. References Aline Migné, François Bordeyne, Dominique Davoult (2023) Long-term survey of intertidal rocky shore macrobenthic community metabolism and structure after primary succession. HAL, ver.2 peer-reviewed and recommended by PCI Ecology https://hal.science/hal-04347756 | Long-term survey of intertidal rocky shore macrobenthic community metabolism and structure after primary succession | Aline Migné, François Bordeyne, Dominique Davoult | <p>Ecological succession involves the transition from opportunistic ephemeral species, which display a minimal variation in functional traits, to slow growing, more functionally diverse, perennial species. The present study aimed in measuring the ... | Biodiversity, Colonization, Community ecology, Ecological successions, Ecosystem functioning, Experimental ecology, Marine ecology | Gudrun Bornette | Thomas Guillemaud, John Griffin, Ignasi Bartomeus, Dilip kumar jha , Abby Gilson , Francisco Arenas, Markus Molis , Matthew Bracken | 2023-12-19 15:39:21 | View | |
16 Dec 2024

From fear to food: predation risk shapes deer behaviour, their resources and forest vegetationJean-Louis Martin, Simon Chamaillé-Jammes, Anne Salomon, Devana Veronica Gomez Pourroy, Mathilde Schlaeflin, Soizic Le Saout, Annick Lucas, Ilham Bentaleb, Simon Chollet, Jake Pattison, Soline Martin-Blangy , Anthony J. Gaston https://hal.science/hal-04381108v5A multidimensional exploration of predator-prey dynamicsRecommended by Gloriana Chaverri based on reviews by Thomas Guillemaud and 2 anonymous reviewers based on reviews by Thomas Guillemaud and 2 anonymous reviewers
In the preprint "From Fear to Food: Predation Risk Shapes Deer Behaviour, Their Resources, and Forest Vegetation", Martin et al. provide a comprehensive examination of the intricate interplay between predation risk, deer behavior, and forest ecosystems. The study offers notable insights into the "ecology of fear," as it takes advantage of an extensive dataset that reflects decades of dedicated research effort. The authors’ approach combines behavioral ecology, plant community analysis, and stable isotope studies, making this work a significant contribution to our understanding of complex ecological phenomena. One of the most striking aspects of this study is the scale and richness of the dataset. The authors used data collected over multiple decades, spanning various experimental contexts, including islands with and without predators, hunting, and culling histories. These datasets are invaluable, as such long-term, geographically diverse studies are rare. The inclusion of both behavioral observations (e.g., flight initiation distances) and ecological outcomes (e.g., vegetation recovery) underscores the effort to provide a holistic understanding of these ecological interactions. The results are not only scientifically robust but also conceptually significant. They challenge simplistic assumptions about predator-prey relationships by illustrating how both the presence and absence of predation risk can have lasting effects on ecosystems. For example, the findings that culling restores vegetation but creates behavioral shifts in deer populations emphasize the complexity of ecological restoration efforts. These results invite further exploration into how behavioral adaptations to predation risk may alter long-term ecosystem trajectories. In conclusion, Martin et al.'s preprint represents a significant advancement in understanding predator-prey interactions and their cascading effects on ecosystems. The study’s comprehensive dataset and integrative approach provide a model for future research in ecological and behavioral sciences. It is a commendable contribution to the field, with implications for both theoretical ecology and practical conservation. References Jean-Louis Martin, Simon Chamaillé-Jammes, Anne Salomon, Devana Veronica Gomez Pourroy, Mathilde Schlaeflin, Soizic Le Saout, Annick Lucas, Ilham Bentaleb, Simon Chollet, Jake Pattison, Soline Martin-Blangy , Anthony J. Gaston (2024) From fear to food: predation risk shapes deer behaviour, their resources and forest vegetation . HAL, ver.6 peer-reviewed and recommended by PCI Ecology https://hal.science/hal-04381108v5 | From fear to food: predation risk shapes deer behaviour, their resources and forest vegetation | Jean-Louis Martin, Simon Chamaillé-Jammes, Anne Salomon, Devana Veronica Gomez Pourroy, Mathilde Schlaeflin, Soizic Le Saout, Annick Lucas, Ilham Bentaleb, Simon Chollet, Jake Pattison, Soline Martin-Blangy , Anthony J. Gaston | <p>The “ecology of fear” posits that predation risk shapes the behaviour of large herbivores, their foraging patterns, their habitat selection and their consequent effect on forest ecology. To test some of these predictions we used the extensive e... |  | Behaviour & Ethology, Biodiversity, Community ecology, Ecosystem functioning, Food webs, Foraging, Habitat selection, Herbivory, Population ecology | Gloriana Chaverri | 2024-01-10 14:07:13 | View |
FOLLOW US
MANAGING BOARD
Julia Astegiano
Tim Coulson
Vasilis Dakos (Representative)
Anna Eklof
Dominique Gravel
François Massol
Ben Phillips
Cyrille Violle










