VERNAY Antoine
- LEHNA, Université Claude Bernard Lyon 1, Lyon, France
- Biodiversity, Biological invasions, Botany, Coexistence, Community ecology, Competition, Ecosystem functioning, Experimental ecology, Facilitation & Mutualism, Foraging, Freshwater ecology, Habitat selection, Interaction networks, Phenotypic plasticity, Terrestrial ecology
- recommender
Recommendations: 2
Review: 1
Recommendations: 2
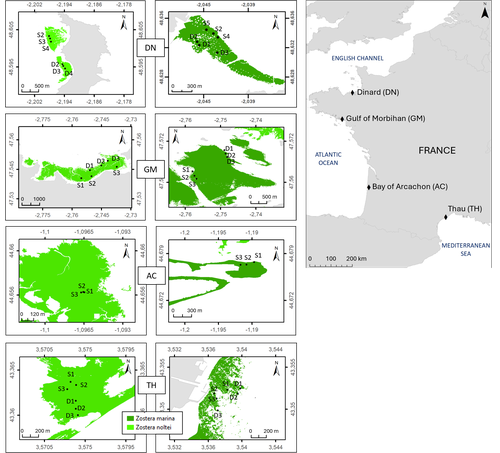
Regional and local variability in the morphometric traits of two emblematic seagrass species (Zostera marina and Zostera noltei) along the French coast
Importance of Scale Considerations in Understanding Seagrass Dynamics
Recommended by Antoine Vernay based on reviews by Gudrun Bornette and 2 anonymous reviewersSeagrasses, particularly Zostera species, have been the subject of numerous studies due to their widespread distribution across the globe (Short et al., 2007), yet they have been in decline for several years as a result of global environmental changes (Touchette, 2007). While case studies and regional studies have been conducted, there remains a lack of information on how to scale these findings, particularly because of the heterogeneous nature of these habitats.
In their study, Lacoste et al. examine the ecosystem trajectories of two Zostera species along a regional gradient spanning sites in the English Channel, the Atlantic Ocean, and the Mediterranean Sea. Their research is based on a recently published database, which offers valuable insights for comparing with other studies and serves as a resource for addressing future questions (Lacoste et al., 2024). This underscores the need for a global database to facilitate the integration of functional responses across studies, thus advancing our understanding of Zostera ecology on a larger scale. The multi-trait approach employed in their study provides a comprehensive view of population dynamics over a 1.5-year period, covering different seasons.
Such studies highlight the complex responses of Zostera populations when considering environmental, seasonal, and geographical heterogeneity. Understanding these dynamics raises important questions about modeling, particularly in relation to the development of a more global database as previously mentioned.
However, the review process has pointed out that the environmental data should be further refined to more rigorously support the presented results. Some statistical analyses could also benefit from improvements to ensure clearer and more explicit conclusions. These concerns are related to the challenges of sampling, the time required for such efforts, and the need to account for spatiotemporal variability. This study could serve as a foundational step for advancing our understanding of Zostera population dynamics on a global scale. In my opinion, despite the large ongoing scientific efforts, upscaling remains one of the major challenges for functional ecologists (Wood et al., 2024), particularly when plant habitats exhibit the kind of heterogeneity seen in Zostera, as demonstrated by Lacoste et al. in their work.
References
Élise Lacoste, Aurélien Boyé, Aline Blanchet-Aurigny, Nicolas Desroy, Isabelle Auby, Touria Bajjouk, Constance Bourdier, Nicolas Cimiterra, Céline Cordier, Amélia Curd, Lauriane Derrien, Élodie Foucault, Jean-Dominique Gaffet, Florian Ganthy, Loic Rigouin, Claire Rollet, Laura Soissons, Aurélien Tancray, Vincent Ouisse (2024) Regional and local variability in the morphometric traits of two emblematic seagrass species (Zostera marina and Zostera noltei) along the French coast. Zenodo, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.5281/zenodo.10427767
Lacoste, E., Ouisse, V., Nicolas, D., Allano, L., Auby, I., Bajjouk, T., Bourdier, C., Caisey, X., de Casamajor, M.-N., Cimiterra, N., Cordier, C., Curd, A., Derrien, L., Droual, G., Dubois, F. S., Foucault, E., Foveau, A., Gaffet, J.-D., Ganthy, F., … Blanchet-Aurigny, A. (2024). A dataset of Zostera marina and Zostera noltei structure and functioning in four sites along the French coast over a period of 18 months. https://doi.org/10.5281/zenodo.14174128
Short, F., Carruthers, T., Dennison, W., & Waycott, M. (2007). Global seagrass distribution and diversity : A bioregional model. Journal of Experimental Marine Biology and Ecology, 350(1), 3‑20. https://doi.org/10.1016/j.jembe.2007.06.012
Touchette, B. W. (2007). The biology and ecology of seagrasses. Journal of Experimental Marine Biology and Ecology, 350(1), 1‑2. https://doi.org/10.1016/j.jembe.2007.06.013
Wood, G. V., Filbee-Dexter, K., Coleman, M. A., Valckenaere, J., Aguirre, J. D., Bentley, P. M., Carnell, P., Dawkins, P. D., Dykman, L. N., Earp, H. S., Ennis, L. B., Francis, P., Franco, J. N., Hayford, H., Lamb, J. B., Ling, S. D., Layton, C., Lis, E., Masters, B., … Wernberg, T. (2024). Upscaling marine forest restoration : Challenges, solutions and recommendations from the Green Gravel Action Group. Frontiers in Marine Science, 11. https://doi.org/10.3389/fmars.2024.1364263
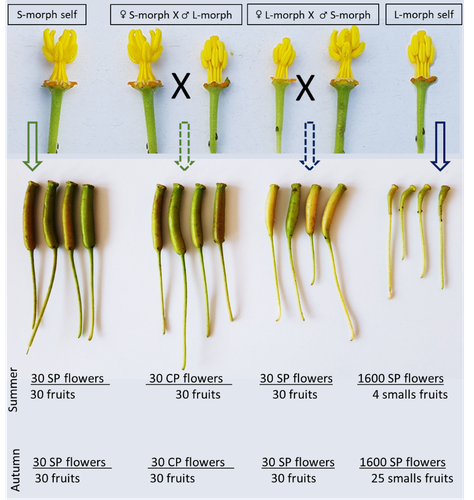
Late-acting self-incompatible system, preferential allogamy and delayed selfing in the heterostylous invasive populations of Ludwigia grandiflora subsp. hexapetala
Water primerose (Ludwigia grandiflora subsp. hexapetala) auto- and allogamy: an ecological perspective
Recommended by Antoine Vernay based on reviews by Juan Arroyo, Emiliano Mora-Carrera and 1 anonymous reviewerInvasive plant species are widely studied by the ecologist community, especially in wetlands. Indeed, alien plants are considered one of the major threats to wetland biodiversity (Reid et al., 2019). Ludwigia grandiflora subsp. hexapetala (Hook. & Arn.) G.L.Nesom & Kartesz, 2000 (Lgh) is one of them and has received particular attention for a long time (Hieda et al., 2020; Thouvenot, Haury, & Thiebaut, 2013). The ecology of this invasive species and its effect on its biotic and abiotic environment has been studied in previous works. Different processes were demonstrated to explain their invasibility such as allelopathic interference (Dandelot et al., 2008), resource competition (Gérard et al., 2014), and high phenotypic plasticity (Thouvenot, Haury, & Thiébaut, 2013), to cite a few of them. However, although vegetative reproduction is a well-known invasive process for alien plants like Lgh (Glover et al., 2015), the sexual reproduction of this species is still unclear and may help to understand the Lgh population dynamics.
Portillo Lemus et al. (2021) showed that two floral morphs of Lgh co-exist in natura, involving self-compatibility for short-styled phenotype and self-incompatibility for long-styled phenotype processes. This new article (Portillo Lemus et al., 2022) goes further and details the underlying mechanisms of the sexual reproduction of the two floral morphs.
Complementing their previous study, the authors have described a late self-incompatible process associated with the long-styled morph, which authorized a small proportion of autogamy. Although this represents a small fraction of the L-morph reproduction, it may have a considerable impact on the L-morph population dynamics. Indeed, authors report that “floral morphs are mostly found in allopatric monomorphic populations (i.e., exclusively S-morph or exclusively L-morph populations)” with a large proportion of L-morph populations compared to S-morph populations in the field. It may seem counterintuitive as L-morph mainly relies on cross-fecundation.
Results show that L-morph autogamy mainly occurs in the fall, late in the reproduction season. Therefore, the reproduction may be ensured if no exogenous pollen reaches the stigma of L-morph individuals. It partly explains the large proportion of L-morph populations in the field.
Beyond the description of late-acting self-incompatibility, which makes the Onagraceae a third family of Myrtales with this reproductive adaptation, the study raises several ecological questions linked to the results presented in the article. First, it seems that even if autogamy is possible, Lgh would favour allogamy, even in S-morph, through the faster development of pollen tubes from other individuals. This may confer an adaptative and evolutive advantage for the Lgh, increasing its invasive potential. The article shows this faster pollen tube development in S-morph but does not test the evolutive consequences. It is an interesting perspective for future research. It would also be interesting to describe cellular processes which recognize and then influence the speed of the pollen tube. Second, the importance of sexual reproduction vs vegetative reproduction would also provide information on the benefits of sexual dimorphism within populations. For instance, how fruit production increases the dispersal potential of Lgh would help to understand Lgh population dynamics and to propose adapted management practices (Delbart et al., 2013; Meisler, 2009).
To conclude, the study proposes a morphological, reproductive and physiological description of the Lgh sexual reproduction process. However, underlying ecological questions are well included in the article and the ecophysiological results enlighten some questions about the role of sexual reproduction in the invasiveness of Lgh. I advise the reader to pay attention to the reviewers’ comments; the debates were very constructive and, thanks to the great collaboration with the authorship, lead to an interesting paper about Lgh reproduction and with promising perspectives in ecology and invasion ecology.
References
Dandelot S, Robles C, Pech N, Cazaubon A, Verlaque R (2008) Allelopathic potential of two invasive alien Ludwigia spp. Aquatic Botany, 88, 311–316. https://doi.org/10.1016/j.aquabot.2007.12.004
Delbart E, Mahy G, Monty A (2013) Efficacité des méthodes de lutte contre le développement de cinq espèces de plantes invasives amphibies : Crassula helmsii, Hydrocotyle ranunculoides, Ludwigia grandiflora, Ludwigia peploides et Myriophyllum aquaticum (synthèse bibliographique). BASE, 17, 87–102. https://popups.uliege.be/1780-4507/index.php?id=9586
Gérard J, Brion N, Triest L (2014) Effect of water column phosphorus reduction on competitive outcome and traits of Ludwigia grandiflora and L. peploides, invasive species in Europe. Aquatic Invasions, 9, 157–166. https://doi.org/10.3391/ai.2014.9.2.04
Glover R, Drenovsky RE, Futrell CJ, Grewell BJ (2015) Clonal integration in Ludwigia hexapetala under different light regimes. Aquatic Botany, 122, 40–46. https://doi.org/10.1016/j.aquabot.2015.01.004
Hieda S, Kaneko Y, Nakagawa M, Noma N (2020) Ludwigia grandiflora (Michx.) Greuter & Burdet subsp. hexapetala (Hook. & Arn.) G. L. Nesom & Kartesz, an Invasive Aquatic Plant in Lake Biwa, the Largest Lake in Japan. Acta Phytotaxonomica et Geobotanica, 71, 65–71. https://doi.org/10.18942/apg.201911
Meisler J (2009) Controlling Ludwigia hexaplata in Northern California. Wetland Science and Practice, 26, 15–19. https://doi.org/10.1672/055.026.0404
Portillo Lemus LO, Harang M, Bozec M, Haury J, Stoeckel S, Barloy D (2022) Late-acting self-incompatible system, preferential allogamy and delayed selfing in the heteromorphic invasive populations of Ludwigia grandiflora subsp. hexapetala. bioRxiv, 2021.07.15.452457, ver. 4 peer-reviewed and recommended by Peer Community in Ecology. https://doi.org/10.1101/2021.07.15.452457
Portillo Lemus LO, Bozec M, Harang M, Coudreuse J, Haury J, Stoeckel S, Barloy D (2021) Self-incompatibility limits sexual reproduction rather than environmental conditions in an invasive water primrose. Plant-Environment Interactions, 2, 74–86. https://doi.org/10.1002/pei3.10042
Reid AJ, Carlson AK, Creed IF, Eliason EJ, Gell PA, Johnson PTJ, Kidd KA, MacCormack TJ, Olden JD, Ormerod SJ, Smol JP, Taylor WW, Tockner K, Vermaire JC, Dudgeon D, Cooke SJ (2019) Emerging threats and persistent conservation challenges for freshwater biodiversity. Biological Reviews, 94, 849–873. https://doi.org/10.1111/brv.12480
Thouvenot L, Haury J, Thiebaut G (2013) A success story: water primroses, aquatic plant pests. Aquatic Conservation: Marine and Freshwater Ecosystems, 23, 790–803. https://doi.org/10.1002/aqc.2387
Thouvenot L, Haury J, Thiébaut G (2013) Seasonal plasticity of Ludwigia grandiflora under light and water depth gradients: An outdoor mesocosm experiment. Flora - Morphology, Distribution, Functional Ecology of Plants, 208, 430–437. https://doi.org/10.1016/j.flora.2013.07.004
Review: 1
A dataset of Zostera marina and Zostera noltei structure and functioning in four sites along the French coast over a period of 18 months
A functional ecology reference database on the populations of two species of Zoostera along french coasts
Recommended by Gudrun Bornette based on reviews by Antoine Vernay, Sara PUIJALON and 1 anonymous reviewerSeagrass beds are in a poor state of conservation and the ecological function of these plant communities is poorly assessed.
Four zones of eelgrass beds (Zostera marina and Zostera noltei) were described in terms of the morphology of the plant populations and the associated fauna. At the same time, parameters related to the functioning of these ecosystems were quantified (benthic fluxes of oxygen, carbon and nutrients) over a two-year cycle.
The article provides the databases collected and provides the main characteristics of these habitats for the measured parameters.
The work provides a reference database on the Zoostera beds of french coastal areas, outlining the ecological contrasts between both ecosystems. This database can on the one hand contribute to help management and restoration of these habitats, and on the other hand provide a reference state of their ecology, with a view to long-term monitoring.
References
Élise Lacoste, Vincent Ouisse, Nicolas Desroy, Lionel Allano, Isabelle Auby, Touria Bajjouk, Constance Bourdier, Xavier Caisey, Marie-Noelle de Casamajor, Nicolas Cimiterra, Céline Cordier, Amélia Curd, Lauriane Derrien, Gabin Droual, Stanislas F. Dubois, Élodie Foucault, Aurélie Foveau, Jean-Dominique Gaffet, Florian Ganthy, Camille Gianaroli, Rachel Ignacio-Cifré, Pierre-Olivier Liabot, Gregory Messiaen, Claire Meteigner, Benjamin Monnier, Robin Van Paemelen, Marine Pasquier, Loic Rigouin, Claire Rollet, Aurélien Royer, Laura Soissons, Aurélien Tancray, Aline Blanchet-Aurigny (2023) A dataset of Zostera marina and Zostera noltei structure and functioning in four sites along the French coast over a period of 18 months.. Zenodo, ver.3 peer-reviewed and recommended by PCI Ecology https://doi.org/10.5281/zenodo.10425140