Direct submissions to PCI Ecology from bioRxiv.org are possible using the B2J service
Latest recommendations

| Id | Title * | Authors * | Abstract * | Picture * | Thematic fields * | Recommender▲ | Reviewers | Submission date | |
|---|---|---|---|---|---|---|---|---|---|
15 Jun 2020

Investigating the rare behavior of male parental care in great-tailed gracklesFolsom MA, MacPherson M, Lukas D, McCune KB, Bergeron L, Bond A, Blackwell A, Rowney C, Logan CJ https://github.com/corinalogan/grackles/blob/master/Files/Preregistrations/gmalecare.RmdStudying a rare behavior in a polygamous bird: male parental care in great-tailed gracklesRecommended by Marie-Jeanne Holveck based on reviews by Matthieu Paquet and André C FerreiraThe Great-tailed grackle (Quiscalus mexicanus) is a polygamous bird species that is originating from Central America and rapidly expanding its geographic range toward the North, and in which females were long thought to be the sole nest builders and caretakers of the young. In their pre-registration [1], Folsom and collaborators report repeated occurrences of male parental care and develop hypotheses aiming at better understanding the occurrence and the fitness consequences of this very rarely observed male behavior. They propose to assess if male parental care correlates with the circulating levels of several relevant hormones, increases offspring survival, is a local adaptation, and is a mating strategy, in surveying three populations located in Arizona (middle of the geographic range expansion), California (northern edge of the geographic range), and in Central America (core of the range). This study is part of a 5-year bigger project. References [1] Folsom MA, MacPherson M, Lukas D, McCune KB, Bergeron L, Bond A, Blackwell A, Rowney C, Logan CJ. 2020. Investigating the rare behavior of male parental care in great-tailed grackles. corinalogan.com/Preregistrations/gmalecare.html In principle acceptance by PCI Ecology of the version on 15 June 2020 corinalogan/grackles/blob/master/Files/Preregistrations/gmalecare.Rmd. | Investigating the rare behavior of male parental care in great-tailed grackles | Folsom MA, MacPherson M, Lukas D, McCune KB, Bergeron L, Bond A, Blackwell A, Rowney C, Logan CJ | This is a PREREGISTRATION submitted for pre-study peer review. Our planned data collection START DATE is May 2020, therefore it would be ideal if the peer review process could be completed before then. Abstract: Great-tailed grackles (Quiscalus... |  | Behaviour & Ethology, Biological invasions, Preregistrations, Zoology | Marie-Jeanne Holveck | 2019-12-05 17:38:47 | View | |
03 Apr 2020
Body temperatures, life history, and skeletal morphology in the nine-banded armadillo (Dasypus novemcinctus)Frank Knight, Cristin Connor, Ramji Venkataramanan, Robert J. Asher https://doi.org/10.17863/CAM.50971Is vertebral count in mammals influenced by developmental temperature? A study with Dasypus novemcinctusRecommended by Mar Sobral based on reviews by Darin Croft and ?Mammals show a very low level of variation in vertebral count, both among and within species, in comparison to other vertebrates [1]. Jordan’s rule for fishes states that the vertebral number among species increases with latitude, due to ambient temperatures during development [2]. Temperature has also been shown to influence vertebral count within species in fish [3], amphibians [4], and birds [5]. However, in mammals the count appears to be constrained, on the one hand, by a possible relationship between the development of the skeleton and the proliferations of cell lines with associated costs (neural malformations, cancer etc., [6]), and on the other by the cervical origin of the diaphragm [7]. References [1] Hautier L, Weisbecker V, Sánchez-Villagra MR, Goswami A, Asher RJ (2010) Skeletal development in sloths and the evolution of mammalian vertebral patterning. Proceedings of the National Academy of Sciences, 107, 18903–18908. doi: 10.1073/pnas.1010335107 | Body temperatures, life history, and skeletal morphology in the nine-banded armadillo (Dasypus novemcinctus) | Frank Knight, Cristin Connor, Ramji Venkataramanan, Robert J. Asher | <p>The nine banded armadillo (*Dasypus novemcinctus*) is the only xenarthran mammal to have naturally expanded its range into the middle latitudes of the USA. It is not known to hibernate, but has been associated with unusually labile core body te... |  | Behaviour & Ethology, Evolutionary ecology, Life history, Physiology, Zoology | Mar Sobral | 2019-11-22 22:57:31 | View | |
14 Jan 2021
Consistent variations in personality traits and their potential for genetic improvement of biocontrol agents: Trichogramma evanescens as a case studySilène Lartigue, Myriam Yalaoui, Jean Belliard, Claire Caravel, Louise Jeandroz, Géraldine Groussier, Vincent Calcagno, Philippe Louâpre, François-Xavier Dechaume-Moncharmont, Thibaut Malausa and Jérôme Moreau https://doi.org/10.1101/2020.08.21.257881Tell us how you can be, and we’ll make you better: exploiting genetic variability in personality traits to improve top-down control of agricultural pestsRecommended by Marta Montserrat based on reviews by Bart A Pannebakker, François Dumont, Joshua Patrick Byrne and Ana Pimenta Goncalves PereiraAgriculture in the XXI century faces the huge challenge of having to provide food to a rapidly growing human population, which is expected to reach 10.9 billion in 2100 (UUNN 2019), by means of practices and methods that guarantee crop sustainability, human health safety, and respect to the environment (UUNN 2015). Such regulation by the United Nations ultimately entails that agricultural scientists are urged to design strategies and methods that effectively minimize the use of harmful chemical products to control pest populations and to improve soil quality. References Bielza, P., Balanza, V., Cifuentes, D. and Mendoza, J. E. (2020). Challenges facing arthropod biological control: Identifying traits for genetic improvement of predators in protected crops. Pest Manag Sci. doi: https://doi.org/10.1002/ps.5857 | Consistent variations in personality traits and their potential for genetic improvement of biocontrol agents: Trichogramma evanescens as a case study | Silène Lartigue, Myriam Yalaoui, Jean Belliard, Claire Caravel, Louise Jeandroz, Géraldine Groussier, Vincent Calcagno, Philippe Louâpre, François-Xavier Dechaume-Moncharmont, Thibaut Malausa and Jérôme Moreau | <p>Improvements in the biological control of agricultural pests require improvements in the phenotyping methods used by practitioners to select efficient biological control agent (BCA) populations in industrial rearing or field conditions. Consist... | Agroecology, Behaviour & Ethology, Biological control, Evolutionary ecology, Life history | Marta Montserrat | 2020-08-24 10:40:03 | View | ||
21 Feb 2019

Photosynthesis of Laminaria digitata during the immersion and emersion periods of spring tidal cycles during hot, sunny weatherAline Migné, Gaspard Delebecq, Dominique Davoult, Nicolas Spilmont, Dominique Menu, Marie-Andrée Janquin and François Gévaert https://hal.sorbonne-universite.fr/hal-01827565v4Evaluating physiological responses of a kelp to environmental changes at its vulnerable equatorward range limitRecommended by Matthew Bracken based on reviews by 2 anonymous reviewersUnderstanding processes at species’ range limits is of paramount importance in an era of global change. For example, the boreal kelp Laminaria digitata, which dominates low intertidal and shallow subtidal rocky reefs in northwestern Europe, is declining in the equatorward portion of its range [1]. In this contribution, Migné and colleagues [2] focus on L. digitata near its southern range limit on the coast of France and use a variety of techniques to paint a complete picture of the physiological responses of the kelp to environmental changes. Importantly, and in contrast to earlier work on the species which focused on subtidal individuals (e.g. [3]), Migné et al. [2] describe responses not only in the most physiologically stressful portion of the species’ range but also in the most stressful portion of its local environment: the upper portion of its zone on the shoreline, where it is periodically exposed to aerial conditions and associated thermal and desiccation stresses. References [1] Raybaud, V., Beaugrand, G., Goberville, E., Delebecq, G., Destombe, C., Valero, M., Davoult, D., Morin, P. & Gevaert, F. (2013). Decline in kelp in west Europe and climate. PloS one, 8(6), e66044. doi: 10.1371/journal.pone.0066044 | Photosynthesis of Laminaria digitata during the immersion and emersion periods of spring tidal cycles during hot, sunny weather | Aline Migné, Gaspard Delebecq, Dominique Davoult, Nicolas Spilmont, Dominique Menu, Marie-Andrée Janquin and François Gévaert | The boreal kelp Laminaria digitata dominates the low intertidal and upper subtidal zones of moderately exposed rocky shores in north-western Europe. Due to ocean warming, this foundation species is predicted to disappear from French coasts in the ... |  | Marine ecology | Matthew Bracken | 2018-07-02 18:03:11 | View | |
06 May 2021

Trophic niche of the invasive gregarious species Crepidula fornicata, in relation to ontogenic changesThibault Androuin, Stanislas F. Dubois, Cédric Hubas, Gwendoline Lefebvre, Fabienne Le Grand, Gauthier Schaal, Antoine Carlier https://doi.org/10.1101/2020.07.30.229021A lack of clear dietary differences between ontogenetic stages of invasive slippersnails provides important insights into resource use and potential inter- and intra-specific competitionRecommended by Matthew Bracken based on reviews by 2 anonymous reviewersThe slippersnail (Crepidula fornicata), originally from the eastern coast of North America, has invaded European coastlines from Norway to the Mediterranean Sea [1]. This species is capable of achieving incredibly high densities (up to several thousand individuals per square meter) and likely has major impacts on a variety of community- and ecosystem-level processes, including alteration of carbon and nitrogen fluxes and competition with native suspension feeders [2]. Given this potential for competition, it is important to understand the diet of C. fornicata and its potential overlap with native species. However, previous research on the diet of C. fornicata and related species suggests that the types of food consumed may change with age [3, 4]. This species has an unusual reproductive strategy. It is a sequential hermaphrodite, which begins life as a somewhat mobile male but eventually slows down to become sessile. Sessile individuals form stacks of up to 10 or more individuals, with larger individuals on the bottom of the stack, and decreasingly smaller individuals piled on top. Snails at the bottom of the stack are female, whereas snails at the top of the stack are male; when the females die, the largest males become female [5]. Thus, understanding these potential ontogenetic dietary shifts has implications for both intraspecific (juvenile vs. male vs. female) and interspecific competition associated with an abundant, invasive species. To this end, Androuin and colleagues evaluated the stable-isotope (d13C and d15N) and fatty-acid profiles of food sources and different life-history stages of C. fornicata [6]. Based on previous work highlighting the potential for life-history changes in the diet of this species [3,4], they hypothesized that C. fornicata would shift its diet as it aged and predicted that this shift would be reflected in changes in its stable-isotope and fatty-acid profiles. The authors found that potential food sources (biofilm, suspended particulate organic matter, and superficial sedimentary organic matter) differed substantially in both stable-isotope and fatty-acid signatures. However, whereas fatty-acid profiles changed substantially with age, there was no shift in the stable-isotope signatures. Because stable-isotope differences between food sources were not reflected in differences between life-history stages, the authors conservatively concluded that there was insufficient evidence for a diet shift with age. The ontogenetic shifts in fatty-acid profiles were intriguing, but the authors suggested that these reflected age-related physiological changes rather than changes in diet. The authors’ work highlights the need to consider potential changes in the roles of invasive species with age, especially when evaluating interactions with native species. In this case, C. fornicata consumed a variety of food sources, including both benthic and particulate organic matter, regardless of age. The carbon stable-isotope signature of C. fornicata overlaps with those of several native suspension- and deposit-feeding species in the region [7], suggesting the possibility of resource competition, especially given the high abundances of this invader. This contribution demonstrates the potential difficulty of characterizing the impacts of an abundant invasive species with a complex life-history strategy. Like many invasive species, C. fornicata appears to be a dietary generalist, which likely contributes to its success in establishing and thriving in a variety of locations [8].
References [1] Blanchard M (1997) Spread of the slipper limpet Crepidula fornicata (L. 1758) in Europe. Current state dans consequences. Scientia Marina, 61, 109–118. Open Access version : https://archimer.ifremer.fr/doc/00423/53398/54271.pdf [2] Martin S, Thouzeau G, Chauvaud L, Jean F, Guérin L, Clavier J (2006) Respiration, calcification, and excretion of the invasive slipper limpet, Crepidula fornicata L.: Implications for carbon, carbonate, and nitrogen fluxes in affected areas. Limnology and Oceanography, 51, 1996–2007. https://doi.org/10.4319/lo.2006.51.5.1996 [3] Navarro JM, Chaparro OR (2002) Grazing–filtration as feeding mechanisms in motile specimens of Crepidula fecunda (Gastropoda: Calyptraeidae). Journal of Experimental Marine Biology and Ecology, 270, 111–122. https://doi.org/10.1016/S0022-0981(02)00013-8 [4] Yee AK, Padilla DK (2015) Allometric Scaling of the Radula in the Atlantic Slippersnail Crepidula fornicata. Journal of Shellfish Research, 34, 903–907. https://doi.org/10.2983/035.034.0320 [5] Collin R (1995) Sex, Size, and Position: A Test of Models Predicting Size at Sex Change in the Protandrous Gastropod Crepidula fornicata. The American Naturalist, 146, 815–831. https://doi.org/10.1086/285826 [6] Androuin T, Dubois SF, Hubas C, Lefebvre G, Grand FL, Schaal G, Carlier A (2021) Trophic niche of the invasive gregarious species Crepidula fornicata, in relation to ontogenic changes. bioRxiv, 2020.07.30.229021, ver. 4 peer-reviewed and recommended by Peer Community in Ecology. https://doi.org/10.1101/2020.07.30.229021 [7] Dauby P, Khomsi A, Bouquegneau J-M (1998) Trophic Relationships within Intertidal Communities of the Brittany Coasts: A Stable Carbon Isotope Analysis. Journal of Coastal Research, 14, 1202–1212. Retrieved May 4, 2021, from http://www.jstor.org/stable/4298880 [8] Machovsky-Capuska GE, Senior AM, Simpson SJ, Raubenheimer D (2016) The Multidimensional Nutritional Niche. Trends in Ecology & Evolution, 31, 355–365. https://doi.org/10.1016/j.tree.2016.02.009
| Trophic niche of the invasive gregarious species Crepidula fornicata, in relation to ontogenic changes | Thibault Androuin, Stanislas F. Dubois, Cédric Hubas, Gwendoline Lefebvre, Fabienne Le Grand, Gauthier Schaal, Antoine Carlier | <p style="text-align: justify;">The slipper limpet Crepidula fornicata is a common and widespread invasive gregarious species along the European coast. Among its life-history traits, well-documented ontogenic changes in behavior (i.e., motile male... |  | Food webs, Life history, Marine ecology | Matthew Bracken | 2020-08-01 23:55:57 | View | |
20 Sep 2018
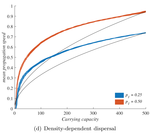
When higher carrying capacities lead to faster propagationMarjorie Haond, Thibaut Morel-Journel, Eric Lombaert, Elodie Vercken, Ludovic Mailleret & Lionel Roques https://doi.org/10.1101/307322When the dispersal of the many outruns the dispersal of the fewRecommended by Matthieu Barbier based on reviews by Yuval Zelnik and 1 anonymous reviewer based on reviews by Yuval Zelnik and 1 anonymous reviewer
Are biological invasions driven by a few pioneers, running ahead of their conspecifics? Or are these pioneers constantly being caught up by, and folded into, the larger flux of propagules from the established populations behind them? References [1] Levins, R., & Culver, D. (1971). Regional Coexistence of Species and Competition between Rare Species. Proceedings of the National Academy of Sciences, 68(6), 1246–1248. doi: 10.1073/pnas.68.6.1246 | When higher carrying capacities lead to faster propagation | Marjorie Haond, Thibaut Morel-Journel, Eric Lombaert, Elodie Vercken, Ludovic Mailleret & Lionel Roques | <p>This preprint has been reviewed and recommended by Peer Community In Ecology (https://dx.doi.org/10.24072/pci.ecology.100004). Finding general patterns in the expansion of natural populations is a major challenge in ecology and invasion biology... |  | Biological invasions, Colonization, Dispersal & Migration, Experimental ecology, Population ecology, Spatial ecology, Metacommunities & Metapopulations, Theoretical ecology | Matthieu Barbier | Yuval Zelnik | 2018-04-25 10:18:48 | View |
10 Jan 2019

Inferring macro-ecological patterns from local species' occurrencesAnna Tovo, Marco Formentin, Samir Suweis, Samuele Stivanello, Sandro Azaele, Amos Maritan https://doi.org/10.1101/387456Upscaling the neighborhood: how to get species diversity, abundance and range distributions from local presence/absence dataRecommended by Matthieu Barbier based on reviews by Kevin Cazelles and 1 anonymous reviewer based on reviews by Kevin Cazelles and 1 anonymous reviewer
How do you estimate the biodiversity of a whole community, or the distribution of abundances and ranges of its species, from presence/absence data in scattered samples? ADDITIONAL COMMENTS 1) To explain the novelty of the authors' contribution, it is useful to look at competing techniques. 2) The main condition for all such approaches to work is well-mixedness: each sample should be sufficiently like a lot drawn from the same skewed lottery. As long as that condition applies, finding the best approach is a theoretical matter of probabilities and combinatorics that may, in time, be given a definite answer. 3) One may ask: why the Negative Binomial as a Species Abundance Distribution? References [1] Fisher, R. A., Corbet, A. S., & Williams, C. B. (1943). The relation between the number of species and the number of individuals in a random sample of an animal population. The Journal of Animal Ecology, 42-58. doi: 10.2307/1411 | Inferring macro-ecological patterns from local species' occurrences | Anna Tovo, Marco Formentin, Samir Suweis, Samuele Stivanello, Sandro Azaele, Amos Maritan | <p>Biodiversity provides support for life, vital provisions, regulating services and has positive cultural impacts. It is therefore important to have accurate methods to measure biodiversity, in order to safeguard it when we discover it to be thre... |  | Macroecology, Species distributions, Statistical ecology, Theoretical ecology | Matthieu Barbier | 2018-08-09 16:44:09 | View | |
10 Jan 2024
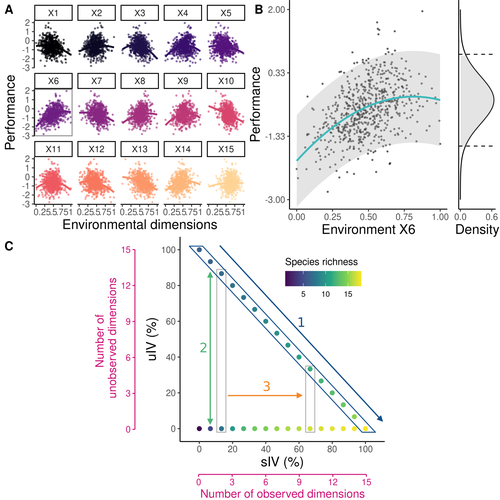
Beyond variance: simple random distributions are not a good proxy for intraspecific variability in systems with environmental structureCamille Girard-Tercieux, Ghislain Vieilledent, Adam Clark, James S. Clark, Benoit Courbaud, Claire Fortunel, Georges Kunstler, Raphaël Pélissier, Nadja Rüger, Isabelle Maréchaux https://doi.org/10.1101/2022.08.06.503032Two paradigms for intraspecific variabilityRecommended by Matthieu Barbier based on reviews by Simon Blanchet and Bart Haegeman based on reviews by Simon Blanchet and Bart Haegeman
Community ecology usually concerns itself with understanding the causes and consequences of diversity at a given taxonomic resolution, most classically at the species level. Yet there is no doubt that diversity exists at all scales, and phenotypic variability within a taxon can be comparable to differences between taxa, as observed from bacteria to fish and trees. The question that motivates an active and growing body of work (e.g. Raffard et al 2019) is not so much whether intraspecific variability matters, but what we get wrong by ignoring it and how to incorporate it into our understanding of communities. There is no established way to think about diversity at multiple nested taxonomic levels, and it is tempting to summarize intraspecific variability simply by measuring species mean and variance in any trait and metric. In this study, Girard-Tercieux et al (2023a) propose that, to understand its impact on community-level outcomes and in particular on species coexistence, we should carefully distinguish between two ways of thinking about intraspecific variability: -"unstructured" variation, where every individual's features are like an independent random draw from a species-specific distribution, for instance, due to genetic lottery and developmental accidents -"structured" variation that is due to each individual encountering a different but enduring microenvironment. The latter type of variability may still appear complex and random-like when the environment is high-dimensional (i.e. multifaceted, with many different factors contributing to each individual's performance and development). Thus, it is not necessarily "structured" in the sense of being easily understood -- we may need to measure more aspects of the environment than is practical if we want to fully predict these variations. What distinguishes this "structured" variability is that it is, in a loose sense, inheritable: individuals from the same species that grow in the same microenvironment will have the same performance, in a repeatable fashion. Thus, if each species is best at exploiting at least a fraction of environmental conditions, it is likely to avoid extinction by competition, except in the unlucky case of no propagule reaching any of the favorable sites. The core intuition, that the complex spatial structure and high-dimensional nature of the environment plays a key explanatory role in species coexistence, is a running thread through several of the authors' work (e.g. Clark et al 2010), clearly inspired by their focus on tropical forests. This study, by tackling the question of intraspecific determinants of interspecific outcomes, makes a compelling addition to this line of investigation, coming as a theoretical companion to a more data-oriented study (Girard-Tercieux et al 2023b). But I believe it raises a question that is even broader in scope. This kind of intraspecific variability, due to different individuals growing in different microenvironments, is perhaps most relevant for trees and other sessile organisms, but the distinction made here between "unstructured" and "structured" variability can likely be extended to many other ecological settings. In my understanding, what matters most in "structured" variability is not so much it stemming from a fixed environment, but rather it being maintained across generations, rather than possibly lost by drift. This difference between variability in the form of "frozen" randomness and in the form of stochastic drift over time is highly relevant in other theoretical fields (e.g. in physics, where it is the difference between a disordered solid and a liquid), and thus, I expect that it is a meaningful distinction to make throughout community ecology. References James S. Clark, David Bell, Chengjin Chu, Benoit Courbaud, Michael Dietze, Michelle Hersh, Janneke HilleRisLambers et al. (2010) "High‐dimensional coexistence based on individual variation: a synthesis of evidence." Ecological Monographs 80, no. 4 : 569-608. https://doi.org/10.1890/09-1541.1 Camille Girard-Tercieux, Ghislain Vieilledent, Adam Clark, James S. Clark, Benoît Courbaud, Claire Fortunel, Georges Kunstler, Raphaël Pélissier, Nadja Rüger, Isabelle Maréchaux (2023a) "Beyond variance: simple random distributions are not a good proxy for intraspecific variability in systems with environmental structure." bioRxiv, ver. 4 peer-reviewed and recommended by Peer Community in Ecology. https://doi.org/10.1101/2022.08.06.503032 Camille Girard‐Tercieux, Isabelle Maréchaux, Adam T. Clark, James S. Clark, Benoît Courbaud, Claire Fortunel, Joannès Guillemot et al. (2023b) "Rethinking the nature of intraspecific variability and its consequences on species coexistence." Ecology and Evolution 13, no. 3 : e9860. https://doi.org/10.1002/ece3.9860 Allan Raffard, Frédéric Santoul, Julien Cucherousset, and Simon Blanchet. (2019) "The community and ecosystem consequences of intraspecific diversity: A meta‐analysis." Biological Reviews 94, no. 2: 648-661. https://doi.org/10.1111/brv.12472 | Beyond variance: simple random distributions are not a good proxy for intraspecific variability in systems with environmental structure | Camille Girard-Tercieux, Ghislain Vieilledent, Adam Clark, James S. Clark, Benoit Courbaud, Claire Fortunel, Georges Kunstler, Raphaël Pélissier, Nadja Rüger, Isabelle Maréchaux | <p>The role of intraspecific variability (IV) in shaping community dynamics and species coexistence has been intensively discussed over the past decade and modelling studies have played an important role in that respect. However, these studies oft... |  | Biodiversity, Coexistence, Community ecology, Competition, Theoretical ecology | Matthieu Barbier | 2022-08-07 12:51:30 | View | |
13 Jul 2020

Preregistration - The effect of dominance rank on female reproductive success in social mammalsShivani, Elise Huchard, Dieter Lukas https://dieterlukas.github.io/Preregistration_MetaAnalysis_RankSuccess.htmlWhy are dominant females not always showing higher reproductive success? A preregistration of a meta-analysis on social mammalsRecommended by Matthieu Paquet based on reviews by Bonaventura Majolo and 1 anonymous reviewer based on reviews by Bonaventura Majolo and 1 anonymous reviewer
In social species conflicts among group members typically lead to the formation of dominance hierarchies with dominant individuals outcompeting other groups members and, in some extreme cases, suppressing reproduction of subordinates. It has therefore been typically assumed that dominant individuals have a higher breeding success than subordinates. However, previous work on mammals (mostly primates) revealed high variation, with some populations showing no evidence for a link between female dominance reproductive success, and a meta-analysis on primates suggests that the strength of this relationship is stronger for species with a longer lifespan [1]. Therefore, there is now a need to understand 1) whether dominance and reproductive success are generally associated across social mammals (and beyond) and 2) which factors explains the variation in the strength (and possibly direction) of this relationship. References [1] Majolo, B., Lehmann, J., de Bortoli Vizioli, A., & Schino, G. (2012). Fitness‐related benefits of dominance in primates. American journal of physical anthropology, 147(4), 652-660. doi: 10.1002/ajpa.22031 | Preregistration - The effect of dominance rank on female reproductive success in social mammals | Shivani, Elise Huchard, Dieter Lukas | <p>Life in social groups, while potentially providing social benefits, inevitably leads to conflict among group members. In many social mammals, such conflicts lead to the formation of dominance hierarchies, where high-ranking individuals consiste... |  | Behaviour & Ethology, Meta-analyses, Preregistrations, Social structure, Zoology | Matthieu Paquet | Bonaventura Majolo, Anonymous | 2020-04-06 17:42:37 | View |
02 Aug 2022

The effect of dominance rank on female reproductive success in social mammalsShivani, Elise Huchard, Dieter Lukas https://doi.org/10.32942/osf.io/rc8naWhen do dominant females have higher breeding success than subordinates? A meta-analysis across social mammals.Recommended by Matthieu Paquet based on reviews by 2 anonymous reviewers based on reviews by 2 anonymous reviewers
In this meta-analysis, Shivani et al. [1] investigate 1) whether dominance and reproductive success are generally associated across social mammals and 2) whether this relationship varies according to a) life history traits (e.g., stronger for species with large litter size), b) ecological conditions (e.g., stronger when resources are limited) and c) the social environment (e.g., stronger for cooperative breeders than for plural breeders). Generally, the results are consistent with their predictions, except there was no clear support for this relationship to be conditional on the ecological conditions. considered As I have previously recommended the preregistration of this study [2,3], I do not have much to add here, as such recommendation should not depend on the outcome of the study. What I would like to recommend is the whole scientific process performed by the authors, from preregistration sent for peer review, to preprint submission and post-study peer review. It is particularly recommendable to notice that this project was a Masters student project, which shows that it is possible and worthy to preregister studies, even for such rather short-term projects. I strongly congratulate the authors for choosing this process even for an early career short-term project. I think it should be made possible for short-term students to conduct a preregistration study as a research project, without having to present post-study results. I hope this study can encourage a shift in the way we sometimes evaluate students’ projects. I also recommend the readers to look into the whole pre- and post- study reviewing history of this manuscript and the associated preregistration, as it provides a better understanding of the process and a good example of the associated challenges and benefits [4]. It was a really enriching experience and I encourage others to submit and review preregistrations and registered reports!
References [1] Shivani, Huchard, E., Lukas, D. (2022). The effect of dominance rank on female reproductive success in social mammals. EcoEvoRxiv, rc8na, ver. 10 peer-reviewed and recommended by Peer Community in Ecology. https://doi.org/10.32942/osf.io/rc8na [2] Shivani, Huchard, E., Lukas, D. (2020). Preregistration - The effect of dominance rank on female reproductive success in social mammals In principle acceptance by PCI Ecology of the version 1.2 on 07 July 2020. https://dieterlukas.github.io/Preregistration_MetaAnalysis_RankSuccess.html [3] Paquet, M. (2020) Why are dominant females not always showing higher reproductive success? A preregistration of a meta-analysis on social mammals. Peer Community in Ecology, 100056. https://doi.org/10.24072/pci.ecology.100056 [4] Parker, T., Fraser, H., & Nakagawa, S. (2019). Making conservation science more reliable with preregistration and registered reports. Conservation Biology, 33(4), 747-750. https://doi.org/10.1111/cobi.13342 | The effect of dominance rank on female reproductive success in social mammals | Shivani, Elise Huchard, Dieter Lukas | <p>Life in social groups, while potentially providing social benefits, inevitably leads to conflict among group members. In many social mammals, such conflicts lead to the formation of dominance hierarchies, where high-ranking individuals consiste... |  | Behaviour & Ethology, Meta-analyses | Matthieu Paquet | 2021-10-13 18:26:42 | View |
MANAGING BOARD
Julia Astegiano
Tim Coulson
Anna Eklof
Dominique Gravel
François Massol
Ben Phillips
Cyrille Violle










